A young Victoria woman with cystic fibrosis who fought to get coverage of a life-changing drug has joined a class-action lawsuit against the government.
Lilia Zaharieva launched a campaign last year after her University of Victoria health plan cut off her access to Orkambi, a drug that can slow disease progression and increase lung function for people with cystic fibrosis.
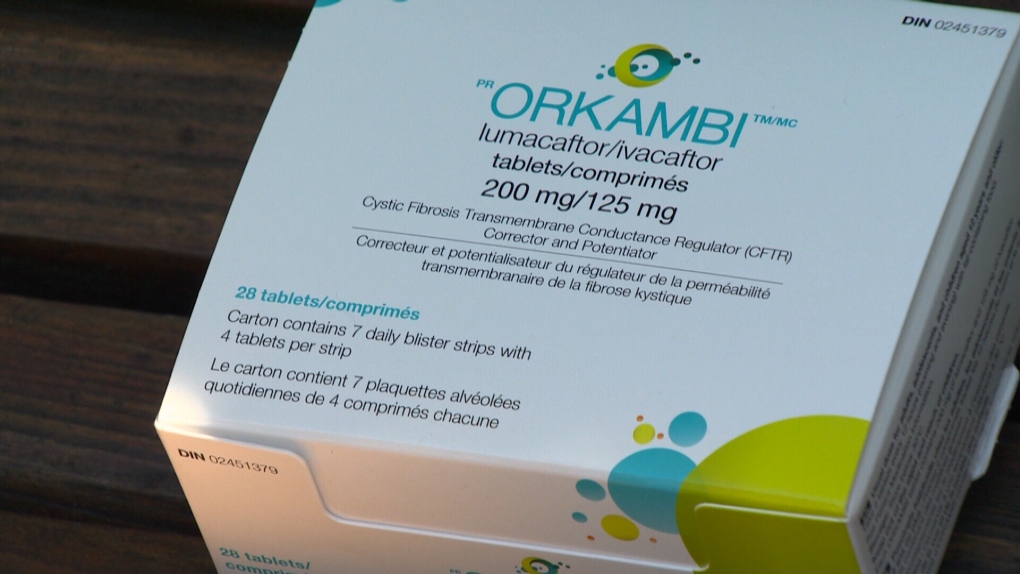
The drug, made by Vertex Pharmaceuticals, comes with a price tag of about $250,000 for a year's supply. It's approved by Health Canada but is not covered under provincial health insurance.
Despite her pleas, the province stood by their decision not to endorse the drug.
Earlier this year the manufacturer offered Zaharieva compassionate access to Orkambi, meaning she wouldn't have to pay the full price.
But her fight to connect others with the medication that changed her life is continuing in a class action lawsuit launched on behalf cystic fibrosis sufferers that is seeking damages of $60-million from the federal and provincial governments, as well as the Canadian Agency for Drugs and Technologies in Health and the Pan Canadian Pharmaceutical Alliance.
The lawsuit says despite evidence-based research showing Orkambi is medically necessary for CF patients, the government is still not covering it under public health insurance plans.
"Section 7 of the Canadian Charter of Rights and Freedoms clearly states that everyone has the right to life, liberty and security of the person and the right not to be deprived thereof except in accordance with the principles of fundamental justice,” said counsel Chris MacLeod. "The Government’s conduct was unlawful in that, the Government’s intentional delegation of policymaking power to CADTH and pCPA was both a violation of the Charter as well as the lack of any underlying lawful authority."
Along with damages of $60-million, the group is requesting a mandatory injunction compelling the provincial government to ensure the availability of Orkambi for patients, and a mandatory injunction for the federal government to institute a transparent system of drug procurement.
"I have had the taste of what life on Orkambi is like. I can’t sit by as others in B.C. wait on the sidelines to gain access," Zaharieva said in a statement. "I’m also subject to losing access to Orkambi at a moment’s notice. The government needs to do the right thing and fix the system now…..not later.”
The Ministry of Health issued a statement in response saying that because it's before the courts, it is unable to comment on specific details of the case.
The ministry did say its current process for approving drug listings is evidence-based and not the result of political lobbying.
"Orkambi is a very expensive drug – with Vertex setting a list cost of about $250,000 per person per year. It is the responsibility of the manufacturer to set a fair, cost-effective and affordable price for their products," the ministry said. "The cost for funding Orkambi for approximately 120 patients in B.C., despite insufficient evidence would be $85.5 million dollars over 3 years. Furthermore, the current price of Orkambi in Canada is under active investigation by the Patented Medicines Price Review Board – Canada’s drug pricing watchdog."
It said the initial recommendation not to cover Orkambi was made in early 2017 under the then-BC Liberal government. Since then, the manufacturer has indicated it has new data about the drug that may affect previous recommendations, according to the ministry.